 < Back
< BackNew publication on cross-border data aggregation
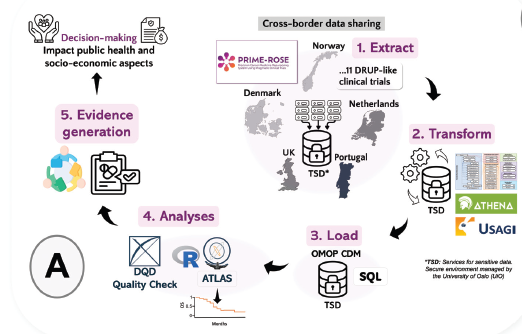
🔎 By pooling data from DRUP-like pragmatic trials across countries, PRIME-ROSE enables faster cohort completion for rare molecular alterations — cohorts that would otherwise remain too small at a national level. Through aligned study designs, harmonized endpoints, and OMOP-based data standardization, we create a shared framework for robust cross-country analyses.
Patients with rare genomic alterations often face limited treatment options and fragmented evidence. Through structured data sharing, OMOP-based harmonization, and aligned statistical analysis plans, PRIME-ROSE enables faster cohort completion and more robust clinical insights across Europe.
Since the launch, PRIME-ROSE has pooled data from 1,000+ patients across 300+ cohorts — demonstrating that collaboration is not just beneficial, but essential,to move the field forward.
🔗Read the full article ‘Crossing borders’ in data standardisation: application of OMOP CDM in an international clinical trial network inprecision cancer medicine


